by Comite Web | Dec 17, 2025 | Sin categorizar
3D-PANC study protocol: Is three-dimensional imaging technology superior to computed tomography for assessing the response to neoadjuvant treatment in patients with borderline or locally advanced pancreatic adenocarcinoma?
This study focuses on improving the preoperative assessment of borderline or locally advanced pancreatic adenocarcinoma (PAC) after neoadjuvant chemotherapy, as conventional computed tomography (CT) has been shown to have very low accuracy in determining which patients will benefit from surgical resection. The hypothesis proposes that applying 3D-MSP (Model for Surgery Planning) technology to CT images will significantly increase accuracy in identifying resectable patients.
The primary objective of the study is to compare the accuracy of 3D-MSP with standard CT, focusing on the diagnosis of post-chemotherapy vascular involvement (venous and/or arterial). To achieve this, a prospective multicenter study is planned that will include, over a one-year period, all patients with PAC undergoing neoadjuvant therapy followed by surgical exploration. The diagnostic accuracy of both techniques (3D-MSP and CT) will be evaluated by comparing their results with intraoperative findings and final histopathological analysis.
Additionally, the study aims to analyze the accuracy of 3D-MSP in demonstrating response to chemotherapy, assess the impact of radiomics (quantitative texture analysis), and validate the usefulness of this three-dimensional technology for education, patient communication, and detailed surgical planning.
Marina Garcés Albir, Francisco García García, Isabel Mora Oliver, Elena Muñoz Forner, Luis Sabater Orti, Dimitri Dorcaratto; GRUPO 3D-PANC Integrantes Grupo 3D-PANC
Department of General and Digestive Surgery, Hospital Clínico Universitario de Valencia, Valencia, Spain; Department of General and Digestive Surgery, School of Medicine, University of Valencia, Valencia, Spain; INCLIVA: Biomedical Research Institute, Valencia, Spain.
Read the full article here!
More about the group
here.
by Comite Web | Dec 16, 2025 | Sin categorizar
The study by Parejo-Alonso et al. identifies PPAR-δ as a critical transcription factor mediating pancreatic cancer cell adaptation to microenvironmental stress.
Normally involved in lipid metabolism, PPAR-δ is overexpressed in pancreatic tumors and, under metabolic stress, senses lipidomic alterations, triggering metabolic reprogramming that enhances glycolysis and promotes a pro-invasive phenotype.
In vitro and in vivo models confirm that pharmacological inhibition of PPAR-δ reverses these adaptations and reduces metastatic potential, highlighting its relevance as a promising therapeutic target for pancreatic cancer treatment.
Complete information at the following link: https://pubmed.ncbi.nlm.nih.gov/40607925/
by Comite Web | Dec 16, 2025 | Sin categorizar
This study conducted by researchers from Vigo presents an innovative sequential therapeutic strategy against pancreatic ductal adenocarcinoma, one of the most lethal cancers due to its stromal barrier that prevents drug penetration.
The novelty lies in the biphasic and sequential treatment: first, lipid nanoparticles containing siRNA that silence the transcription factors YAP1 and FOSL1 are administered, weakening the tumor’s protective stroma; then combination chemotherapy (entinostat and gemcitabine) is applied, which penetrates more effectively into the now permeable tumor tissue.
Results in preclinical models (mouse xenografts and patient organoids) demonstrated reduced fibrotic tissue, increased drug penetration and superior therapeutic efficacy. This approach represents a paradigm shift from traditional combination therapies.
Immacolata Maietta, Patricia Domínguez Arístegui, Iban González Álvarez, Omayra Rodríguez Atanes, Eva María García Fontán, Susana Teijeira Bautista, África González-Fernández, Rosana Simón-Vázquez, Sequential YAP1/FOSL1 silencing and epigenetic therapy to overcome stromal barriers in pancreatic cancer, International Journal of Pharmaceutics, 2025, vol. 684, 126155. https://doi.org/10.1016/j.ijpharm.2025.126155.
Access the full article at the following link: https://pdf.sciencedirectassets.com/271189/1-s2.0-S0378517325X00156/1-s2.0-S0378517325009925/main.pdf?X-Amz-Security-Token=IQoJb3JpZ2luX2VjEKX%2F%2F%2F%2F%2F%2F%2F%2F%2F%2FwEaCXVzLWVhc3QtMSJHMEUCIEmy%2FlDACu%2BJwqxPbPi%2B%2FdT8L3F3qWlzvnC2iVZ8xzfLAiEAiQlAXbjBadOgjBg8Wl%2BrHpj2eQpEWLzo4KCl6C6r4eAqswUIbhAFGgwwNTkwMDM1NDY4NjUiDN1865%2B9jlmTrQV6mSqQBaBj4CJJhNs7rR9%2FaKngxlEdVyp3ciVQDZ86XZQRWnw%2FonwJX3RAqszW3YWaaFbzNVdRDwsWyEqwvnldCO3fKo%2BZZmH%2Fw4zIkdGCq2OSY31%2Bv3lpio2zYlE6Rq8su75k1ZreitM81AmUfjbJB%2FCNwFoFaREMHgtGqWJKCx0%2FeTc8pofkmPmmpbm%2BF%2Bu3JdrtXZ5Zt885mqylBP0zXDovPhguqx%2BpQT8hzMhSRU20mx7Z5P6UM0VpCKx%2BGYHXqN6%2BbEyiybTi1Q2iFPG7t%2BUjpOZEKZZLdezVm%2BC9O2Q0FVzgsC63ffGKPCLS%2BH2hUdfSxzSWq9ij14MiH2h1jlsjGdw4xUDuL4uXesK5ezbyy6JT6%2FH46QdNZvitErBc5d8cfkNHhL0I%2F0GARzLYyOkjhFlZBCjKiH83gkLH0wEjSo2k5aHOLr4Ts49WrF9k8vL5fXEGO7fSSjPCnaV8DB43SO0l6vPHX%2BydavJF59AvMtuijHUxs6wmgzBulOsphtep2V5NDexVilR9IhpJtHOMR15876hFAF%2By3W7W%2FhjuHsnRaTQ3Rw7VHMQB%2BNGzd9JdZ5BQKjh%

by Comite Web | Dec 15, 2025 | Sin categorizar
by Comite Web | Dec 10, 2025 | Sin categorizar
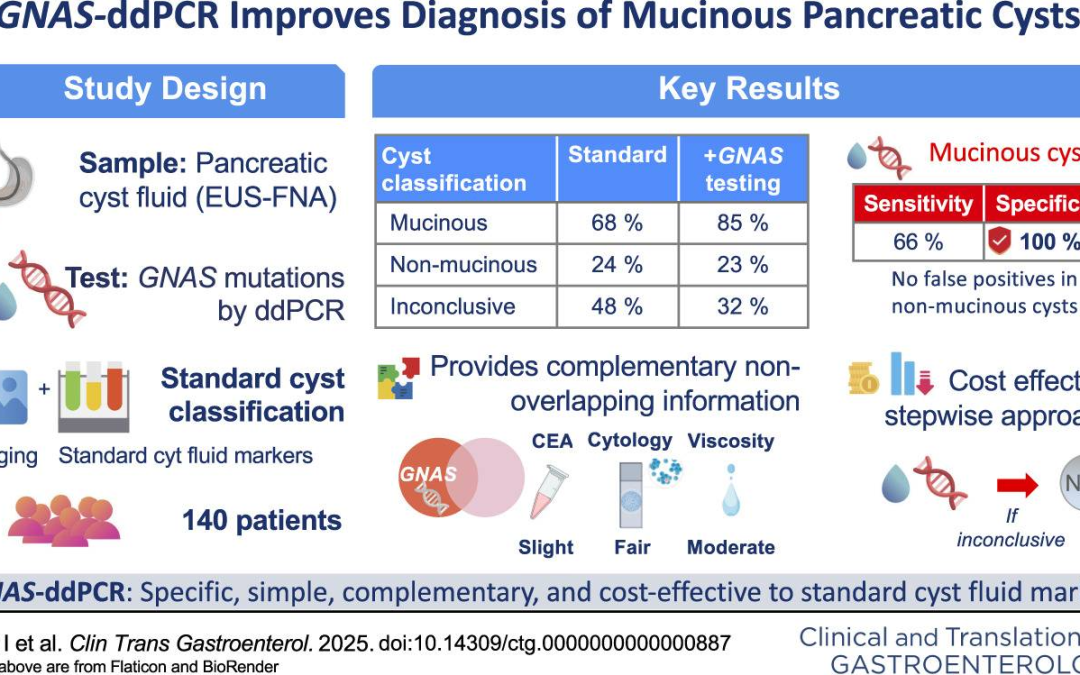
The analysis of GNAS mutations using ddPCR in fluid obtained through endoscopic ultrasound–guided fine-needle aspiration has proven to be a useful and cost-effective diagnostic tool for evaluating mucinous pancreatic cystic neoplasms.
This technique can be applied before turning to more expensive multigene sequencing panels, which can be reserved for cases in which no GNAS mutations are detected and alternative non-mucinous etiologies need to be explored.
Overall, this strategy represents a practical advance that improves diagnostic accuracy and optimizes resource use in the assessment of pancreatic cystic neoplasms.
Araujo IK, Soy G, Ginès A, Sendino O, Fernández-Esparrach G, Sánchez-Montes C, Cuatrecasas M, Archilla I, Montironi C, Silvia A, Ausania F, Domínguez-Fraile M, Villagrasa V, López-Guerra M, Colomer D, Vaquero EC. Single GNAS Droplet-Based Digital Polymerase Chain Reaction Analysis of Pancreatic Cyst Fluid: An Effective Up-Front Strategy for Mucinous Cyst Diagnosis by Endoscopic Ultrasound-Guided Fine-Needle Aspiration.Clin Transl Gastroenterol. 2025 Sep 1;16(9):e00887.
Link to PDF: https://pmc.ncbi.nlm.nih.gov/articles/PMC12456567/pdf/ct9-16-e00887.pdf
Link to the group web: https://alipanc.org/grupo/pancreas-idibaps-clinic/

by Comite Web | Dec 10, 2025 | Sin categorizar
A study led by the Experimental Oncology Group at the Spanish National Cancer Research Centre (CNIO) has described a triple-combination therapy for pancreatic cancer. The work has been published in the prestigious journal Proceedings of the National Academy of Sciences (PNAS).
The study shows that genetic ablation of three key nodes in KRAS signaling (RAF1, EGFR, and STAT3) leads to complete and permanent regression of orthotopic PDAC tumors driven by KRAS/TP53 mutations. Similarly, a drug combination targeting KRAS (RMC-6236/daraxonrasib), EGFR (afatinib), and STAT3 (SD36) induces full tumor regression with no resistance observed for over 200 days. This triple therapy proved effective in both genetically engineered mouse models and patient-derived xenografts, without tumor relapse. Importantly, the treatment was well tolerated. Although the findings support the development of clinical trials based on this combined strategy to improve PDAC therapy, alternatives to afatinib and SD36 will be needed to facilitate clinical translation by reducing toxicity and improving ADME properties, respectively.
This finding opens the door to exploring these therapeutic avenues and guiding the development of new clinical trials that could benefit patients with pancreatic ductal adenocarcinoma.
Link to ➡️ Full article
Links to Research group ➡️ Experimental Oncology ALIPANC, Experimental Oncology CNIO.




